School of Dirk Valkenburg [Public domain], via Wikimedia Commons
Recent studies have sparked a controversy by suggesting that an unusually large number of African Americans possess a gene that is linked with violent behavior. The gene contributes to the regulation of the amounts of monoamine oxidase in the individual’s brain. This enzyme affects levels of neurotransmitters that are known to play an important role in modulating emotion and behavior, such as dopamine, serotonin and norepinephrine.
There are different versions of this gene, and each version contributes to differences in the amount of monoamine oxidase the individual’s brain is likely to have. This is because the difference in these versions has to do with differences in the number of times a specific section for its promoter region appears. The three repeat alleles lead to unusually low levels of the enzyme, and the two repeat alleles lead to lower levels yet.
The early 1990s saw an explosion in the research of the role monoamine oxidase plays in the brain after the study of a family with a rare mutation had an extremely high number of violent criminals. The mutation this family had led to very low levels of monoamine oxidase and low IQs. It came to be known as “Brunner’s syndrome” after H.G. Brunner, the researcher who discovered the gene. Along with his team of researchers, they authored the paper “X-Linked Borderline Mental Retardation with Prominent Behavioral Disturbance: Phenotype, Genetic Localization, and Evidence for Disturbed Monoamine Metabolism.”
They summarized their findings as follows:
“We have identified a large Dutch kindred with a new form of X-linked nondysmorphic mild mental retardation. All affected males in this family show very characteristic abnormal behavior, in particular aggressive and sometimes violent behavior. Other types of impulsive behavior include arson, attempted rape, and exhibitionism. Attempted suicide has been reported in a single case. The locus for this disorder could be assigned to the Xpll-21 interval between DXS7 and DXS77 by linkage analysis using markers spanning the X chromosome. A maximal multipoint lod score of 3.69 was obtained at the monoamine oxidase type A (MAOA) locus in Xpll.23-11.4. Results of 24-h urine analysis in three affected males indicated a marked disturbance of monoamine metabolism. These data are compatible with a primary defect in the structural gene for MAOA and/or monoamine oxidase type B (MAOB). Normal platelet MAOB activity suggests that the unusual behavior pattern in this family may be caused by isolated MAOA deficiency.”
Researchers were later able to genetically engineer mice with an artificial version of this gene, and these rats were likewise more aggressive than other rats. In 1995, Cases and colleagues:
“Deficiency in monoamine oxidase A (MAOA), an enzyme that degrades serotonin and norepinephrine, has recently been shown to be associated with aggressive behavior in men of a Dutch family. A line of transgenic mice was isolated in which transgene integration caused a deletion in the gene encoding MAOA, providing an animal model of MAOA deficiency. In pup brains, serotonin concentrations were increased up to ninefold, and serotonin-like immunoreactivity was present in catecholaminergic neurons. In pup and adult brains, norepinephrine concentrations were increased up to twofold, and cytoarchitectural changes were observed in the somatosensory cortex. Pup behavioral alterations, including trembling, difficulty in righting, and fearfulness were reversed by the serotonin synthesis inhibitor parachlorophenylalanine. Adults manifested a distinct behavioral syndrome, including enhanced aggression in males.”
Numerous other researchers replicated these findings in rats in 2002, 2007, 2008. A breakthrough emerged in 2002 when Caspi and colleagues find that individuals with a 2R or 3R version of the gene exhibited aggression, but only when they had difficult upbringings. They found that it is therefore very important to examine the complex way socioeconomic status and genes interact with one another. Clearly, it is not a “nature vs. nurture” issue, but as is often the case, a question of a very complicated interaction between the two. Bryd and Manuch found this in 2013 as well in a meta-analysis, in which 27 studies on the subject were collated in “MAOA, Childhood Maltreatment, and Antisocial Behavior: Meta-analysis of a Gene-Environment Interaction”; 20 of these cohorts were found to replicate the effects.
“Background
In a seminal study of gene-environment interaction, childhood maltreatment predicted antisocial behavior more strongly in male subjects carrying an MAOA promoter variant of lesser, compared with higher, transcriptional efficiency. Many further investigations have been reported, including studies of other early environmental exposures and female subjects. Here, we report a meta-analysis of studies testing the interaction of MAOA genotype and childhood adversities on antisocial outcomes in predominantly nonclinical samples.
Methods
Included were 27 peer-reviewed, English-language studies published through August, 2012, that contained indicators of maltreatment or other family (e.g., parenting, sociodemographic) hardships; MAOA genotype; indices of aggressive and antisocial behavior; and statistical test of genotype-environment interaction. Studies of forensic and exclusively clinical samples, clinical cohorts lacking proportionally matched control subjects, or outcomes nonspecific for antisocial behavior were excluded. The Liptak-Stouffer weighted Z-test for meta-analysis was implemented to maximize study inclusion and calculated separately for male and female cohorts.
Results
Across 20 male cohorts, early adversity presaged antisocial outcomes more strongly for low-activity, relative to high- activity, MAOA genotype (p = .0044). Stratified analyses showed the interaction specific to maltreatment (p = .00000082) and robust to several sensitivity analyses. Across 11 female cohorts, MAOA did not interact with combined early life adversities, whereas maltreatment alone predicted antisocial behaviors preferentially, but weakly, in female subjects of high-activity MAOA genotype (p = .02).
Conclusions
We found common regulatory variation in MAOA to moderate effects of childhood maltreatment on male antisocial behaviors, confirming a sentinel finding in research on gene-environment interaction. An analogous, but less consistent, finding in female subjects warrants further investigation.”
Thus, this effect is consistent in male subjects, and somewhat prevalent in abused female subjects, though the latter finding is less consistent. Some studies have linked low-activity MAOA to obesity and credit card debt as well. Roux 2014 , Vaughn et al. 2009 , Beaver et al. 2009 , and Beaver et al. 2013 have all found that possession of low-activity MAOA genes increase the risk that the individual will end up in prison. He writes in “Exploring the association between the 2-repeat allele of the MAOA gene promoter polymorphism and psychopathic personality traits, arrests, incarceration, and lifetime antisocial behavior”:
“A line of research has revealed that a polymorphism in the promoter region of the MAOA gene is related to antisocial phenotypes. Most of these studies examine the effects of low MAOA activity alleles (2-repeat and 3-repeat alleles) against the effects of high MAOA activity alleles (3.5-repeat, 4-repeat, and sometimes 5-repeat alleles), with research indicating that the low MAOA activity alleles confer an increased risk to antisocial phenotypes. The current study examined whether the 2-repeat allele, which has been shown to be functionally different from the 3-repeat allele, was associated with a range of antisocial phenotypes in a sample of males drawn from the National Longitudinal Study of Adolescent Health. Analyses revealed that African-American males who carried the 2-repeat allele were, in comparison with other African-American male genotypes, significantly more likely to be arrested and incarcerated. Additional analyses revealed that African-American male carriers of the 2-repeat allele scored significantly higher on an antisocial phenotype index and on measures assessing involvement in violent behaviors over the life course. There was not any association between the 2-repeat allele and a continuously measured psychopathic personality traits scale. The effects of the 2-repeat allele could not be examined in Caucasian males because only 0.1% carried it.”
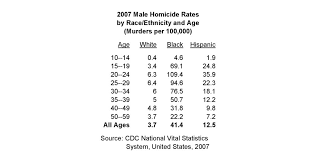
Controversially, there is a great deal of race-differential in the distribution of these genes. In one study of 2,574 whites and 164 blacks, Beaver et al. (2013) found that 5.2 of the black subjects possessed the highest risk 2R allele whereas only 0.1 percent of whites had it. Widom and Brzustowicz (2006) in a study of 180 whites and 98 non-whites found that 6.2 percent of the non-whites had it and 0.6 percent of whites had it. In one study of psychiatric patients, Reti et al. (2011) found that 4.7 percent of 232 blacks had the gene whereas only 0.5 percent of the 232 whites had it. Choe et al. (2014) found that 6 percent of 86 blacks had it whereas 0 percent of 106 whites had it.
The same thing has been found for the 3R version of the gene. This gene is still associated with increased violence, but not as severely as the 2R gene, which causes lower levels of monoamine oxidase. Sabo, Hu and Hamer (1998) found among 539 whites and 52 blacks that 59.1 percent of blacks had it and 33.1 percent of whites had it. Beaver et al. (2013) found that 52.3 percent of blacks had it and 37 percent of whites had it among 2,574 whites and 164 blacks. Widom and Brzustowicz (2006) found 74.1 percent among blacks and 62.8 percent among whites among 196 whites and 143 non-whites. Rosenberg et al. (2006) found 54.1 percent of blacks and 36.1 percent of whites among 37 blacks and 274 whites. Reti et al. found 48.7 percent of blacks and 34.1 of whites among 365 whites and 232 blacks. Choe et al. found 42.2 percent blacks and 30 whites among 106 whites and 83 blacks.
https://www.tremr.com/Duck-Rabbit/african-americans-possess-violence-gene-researchers-find